Sleep Physiology
The human brain exists in three primary states: wake, sleep with rapid eye movements (REM), and sleep without rapid eye movements (NREM). While sleep clearly subserves an essential role, so much remains unexplained about this physiologic state. Since the discovery of REM in the 1950s, there has been much interest in the psychiatric and neurologic communities to develop a better understanding of the physiologic underpinnings and neurobehavioral correlates of sleep. Even though each of the states of sleep serve a vital role in the maintained function of all animals, as demonstrated by the physical deterioration and eventual death that some animals experience with sleep deprivation, there remain vast deficits in the fundamental understanding of sleep’s purpose.
Sleep Architecture
Three physiological sleep parameters should be analyzed in order to be able to describe sleep architecture (structure):
- Signals generated from the cerebral cortex (registered by electroen-cephalogram [EEG]) Electrode placement for EEG scoring rules follows the international 10-20 system, which assigns a number to each EEG electrode to specify the loca¬tion in the left or right hemisphere
- Eye movements (registered by electro-oculogram [EOG]), electrode placement near the eye at the inner and outer canthus
- Face muscles activity (picked up by submandibular muscles electromyography [EMG])
The stages of sleep are when the electrical activity and physiological responses, in the brain, changes through the many periods of sleep. There are five stages of sleep. Scientists categorized the stages of sleep based on the characteristics of the brain and body during sleep. Stage 1,2,3, and 4, are categorized as ‘non-REM sleep’, and the fifth stage, is REM sleep. Generally, brainwave frequencies and amplitudes from an electroencephelogram (EEG) are used to differentiate the different stages of sleep, along with other biologic rhythms including eye movements (EOG) and muscle movements (EMG).
The Five Stages of Sleep
Non-REM Sleep
According to the American Academy of Sleep Medicine NREM is divided into 3 stages defined as N1, N2, N3, where N1 means the shallowest and N3 the deepest sleep.
Stage 1 Sleep (N1)
Stage 1 is the lightest stage of sleep. The EEG brain frequency is slightly slower than during wake time. There is muscle tone present in the skeletal muscles. Breathing occurs at a regular rate.
Stage 2 Sleep (N2)
Stage 2 usually follow Stage 1 and represents deeper sleep. During Stage 2 sleep, the sleeper is less able to be awakened. Stage 2 sleep is characterized by ‘saw tooth waves’ and sleep spindles.
Stage 3 and 4 Sleep (N3)
Stage 3 and Stage 4 sleep are progressively deeper stages of sleep. These stages of sleep are also called ‘Slow Wave Sleep’ (SWS), or delta sleep. During SWS, the EEG shows a much slower frequency with high amplitude signals (delta waves). A sleeper in SWS is often difficult to awaken. Some studies have demonstrated that very loud noises, sometimes over 100 decibels, will not awaken some during SWS. As humans get older they spend less time in slow wave deep sleep and more time in Stage 2 sleep.
Slow-wave sleep is generally referred to as deep sleep, and is comprised of the deepest stage of NREM. In stage three we see the greatest arousal thresholds, such as difficulty in awakening.
After being awoken, the person will generally feel quite groggy, and cognitive tests that have been administered after being awoken from the third stage show that for up to half an hour or so, and when compared to awakenings from the other stages, mental performance is moderately impaired. This is a phenomenon known as sleep inertia. When sleep deprivation has occurred there’s generally a sharp rebound of slow-wave sleep, which suggests that there’s a need for slow-wave sleep. It now appears that slow-wave sleep is a highly active state, and not a brain quiescence as previously believed. In fact, brain imaging data shows that regional brain activity during non-REM sleep is influenced by the most recent waking experience.
Stage 5 Sleep (REM Sleep)
Stage 5, or REM sleep, is the stage of sleep associated with dreaming. It is very different physiologically from the other stages of sleep. The EEG resembles wake time. However, the skeletal muscles are atonic, or without movement. The breathing is more erratic and irregular. The heart rate often increases. It is theorized that muscle atonia evolved in order to protect the individual from injury during sleep. REM Sleep Plays a Prominent Role in Memory Formation.
Stages of sleep characteristics: K-Complexes and Sleep Spindles

Spindle activity is exclusive to NREM sleep, with most occurring at the start and end of NREM. Sleep spindles engage brain activation in the superior temporal gyri, anterior cingulate, insular cortices and the thalamus. Sleep spindles have different lengths; with slow spindles associated with an increase in activity in the area known as the superior frontal gyrus ranging between 11 and 13 Hz, and fast spindles associated with the recruitment of both the hippocampus and the mesial frontal cortex and the sensorimotor processing cortical regions ranging between 13 and 15 Hz. At this point in time it’s not clear what is meant by these sleep spindles, but it’s hoped that ongoing research will reveal their function.
Defining K-Complexes
K-Complexes are also exclusive to NREM sleep, and can be defined as single long delta waves lasting for only a second. Like sleep spindles, they appear automatically during the early stages of sleep, generally in stage two. However, K-Complexes can be induced at will by momentary noises, such as someone knocking on a door. Further research must be conducted on K-Complexes because their function is currently unknown.
Dreaming during NREM
During REM sleep, study participants reported both intense dream vividness and improved memory of dreams which occurred during that phase, which suggests that dreaming typically occurs REM sleep: we know that dreaming also occurs during NREM sleep; however, in comparison, these dreams appear to be more mundane. We also know that dreams that occur during the NREM stage of sleep typically occur after midnight, which happens to be the time-period with the highest rate of REM sleep. This was discovered by a study whereby subjects took naps over specific periods of time and were then forcefully woken: sleep was divided into – naps with only REM sleep, and naps with only NREM sleep using polysomnography.
Factors Influencing Sleep Architecture
Genetic Factors
Researchers in the Netherlands identified seven genes linked with insomnia, meaning there's a biological component to the sleep disorder. These seven genes could indicate that a person has an increased risk for the sleep disorder. One of the genes that the researchers identified was linked to other sleep disorders in previous studies. One of these sleep disorders is a condition called periodic limb movement disorder, involves repetitive movements of the arms and legs during sleep, and the other is called restless legs syndrome, which is characterized by strange and often painful sensations in the legs and an irresistible urge. to move them while resting.
The researchers also noted that there was some overlap between the genes linked to insomnia and the genes linked to other mental health conditions, such as anxiety, depression and neuroticism. "This is an interesting finding, because these characteristics tend to go hand in hand with insomnia," study co-author Anke Hammerschlag, a doctoral student of functional genomics at Vrije Universiteit Amsterdam, said in a statement. "We now know that this is partly due to the shared genetic basis."
How Long You Sleep May Be in Your Genes
The amount of time people spend sleeping is linked with two regions of their DNA, a new study suggests. In the study, researchers examined data from more than 47,000 people of European ancestry who were participating in ongoing studies in Europe, the United States and Australia, and nearly 5,000 African-Americans. The researchers compared people's genetic information with how long they reported sleeping on an average night.
The results revealed two regions of DNA that might be related to how long a person usually sleeps. The first of the two regions was associated with longer-than-average sleeping times, the new study showed. In previous research, this region has also been linked with better glucose metabolism and a lower likelihood of attention deficit hyperactivity disorder. The other region was associated with shorter-than-average sleeping times, and previous studies had linked it with an increased risk of depression and schizophrenia.
"Sleep patterns are influenced by genetic differences," said study author Dr. Daniel Gottlieb, the director of the Sleep Disorders Center at VA Boston Healthcare System. "This study is one of the first to begin identifying these genetic differences, and will hopefully help us better understand the causes of sleep disorders and their relation to other important conditions, such as diabetes and psychiatric disorders."
Age Impact on Sleep
Over a typical lifespan, the amount of time we spend each day sleeping declines. Newborns spend from 16 to 20 hours asleep each day. Between the ages of one and four, total daily sleep time decreases to about 11 or 12 hours. This gradual decline continues through childhood, such that an adolescent will need - though not necessarily get - about nine hours of sleep to function at his or her best. Adults through middle age need at least eight hours, and although the elderly may still require up to eight hours, they may struggle to obtain those hours in one block.
In addition to changes in sleep duration, sleep patterns also change as we age. In the beginning, as all new parents discover, a newborn's sleep is sporadic: the need to sleep and the need to eat cycle across the day and night, with little time for anything else. After three or four months, infants begin to develop a pattern in which sleep becomes consolidated into longer periods. Older infants and young children typically obtain their sleep during a solid nighttime session plus two or more daytime naps. Generally speaking, through the toddler years, naps become fewer in number and shorter in duration, and sleep becomes more consolidated during the night. By the age of six or seven, many children have stopped taking naps entirely. Their sleep is experienced much as it will be through adulthood: in a single consolidated block, most often at night.
A number of sleep problems are particular to adult women. Half of them report sleep disturbances during their menstrual periods; three-quarters of expectant mothers report that sleep is more disturbed during pregnancy; and many experience disrupted sleep during menopause, in part due to nighttime "hot flashes."
Insomnia and disrupted sleep in elderly people are a common side effect caused by many chronic medical conditions such as arthritis, congestive heart failure, depression, and gastroesophogeal reflux disorder. Respiratory disorders, such as sleep apnea, which cause multiple arousals during the night, also become more common as people age. Other problems, such as restless legs syndrome, which results in an uncontrollable need to move one's legs while drifting off to sleep, or periodic limb movements, which cause jerking of the feet or legs during the night, can make it difficult to fall asleep or lead to highly fragmented sleep. Unfortunately, sleep problems in older adults often go undiagnosed and untreated simply because many people believe sleep problems are a normal part of aging or that nothing can be done to help them sleep better. Thankfully, treating any underlying medical disorders can dramatically improve sleep.
Because most older adults are less able than younger adults to maintain sleep, the elderly suffer disproportionately from chronic sleep deprivation. Sleep deprivation may cause individuals to unintentionally nod off during daytime activities. Late afternoon naps can also reduce a person’s ability to sleep through the night, thus potentially worsening insomnia. Learning how the internal clock and sleep drive interact, and how they limit when good sleep can occur, can help people devise strategies that will help them maintain quality sleep as they age.
Sleep Regulation
Neurobiological processes that regulate sleep are very complex. The most important neurotransmitters connected to the sleep regulation are:

- Gamma-Aminobutyric Acid (GABA) system – sleeping and kalming (tranquilizes) pills promote sleep through neurons of this system.
- Histamine system – plays essential role not only in sleep regulation but also in allergic and inflammatory reactions. Via this system allergic and inflammatory reaction deteriorate sleep and on the other hand anti histaminic preparations promote sleep and may cause somnolence within day-time.
- Serotonin system – plays essentials role in deep sleep promotion and inhibition of REM sleep. Serotonin deficiency is responsible for sleep disorders in course of depression. Through this system act several antidepressants promoting sleep and its depth.
- Norepinephrine system – excessive activity of this system and poor sleep quality is often observed in stressful people, people with anxiety, and people suffering from circulatory system diseases e.g. hypertension.
- Cholinergic system – cholinergic neurons produce acetylcholine, promote REM sleep and are connected to memory processes. Deterioration of their function and reduction of REM sleep amount is observed in patients with memory disorders/problems and dementia patients (Alzheimer type) in particular.
- Melatonin system – function of this system is related to the circadian (daylight) rhythm affecting internal biological clock is located in front hypothalamus and suprachiasmatic nuclei* (SCN). The internal biological clock regulates melatonin secretion by pituitary gland.
- Hypocretin/orexin system – it stabilizes sleep regulatory processes. Orexin is a neuropeptide that regulates arousal, wakefulness, and appetite. The most common form of narcolepsy, in which the sufferer experiences brief losses of muscle tone (cataplexy), is caused by a lack of orexin in the brain due to destruction of the cells that produce it.
- Adenosine system – promotes sleep and causes somnolence, which is the more intense the longer is time from last sleep/nap. The adenosine somnolence could be partially blocked by caffeine.
* SCN is a tiny region of the brain in the hypothalamus, situated directly above the optic chiasm. It is responsible for controlling circadian rhythms.

The above list provides some most important sleep regulating agents only. To understand all discovered agents/substances their impact on sleep and relationships between them is pretty difficult and still not fully revealed. The people suffering from sleep disturbances should acquire some knowledge how to care own sleep quality. It would be worth to learn some practical models explaining sleep regulation. One of the most important is two-process model of sleep regulation proposed by professor Alexander Borbély Swiss neurophysiologist of Hungarian descent in early 1980s.
Two-process model of sleep regulation

The sleep-wake cycle, is regulated by two separate biological mechanisms in the body, which interact together and balance each other. It is often referred to as the two-process model of sleep-wake regulation. The two processes are:
- Circadian rhythm, also known as Process C, the regulation of the body’s internal processes and alertness levels (including the circadian drive for arousal and circadian alerting system), which is governed by the internal biological or circadian clock
- Sleep-wake homeostasis, or Process S, the accumulation of hypnogenic (sleep-inducing) substances in the brain, which generates a homeostatic sleep drive.
Both of these processes are influenced to some extent by the genes of the individual. In addition, various external factors (e.g. food, drugs, ambient temperature, meal times, naps, stress, exercise, daily schedules, alarm clocks, etc) can also have a direct or indirect effect on an individual’s sleep-wake cycle.
The body’s built-in circadian clock, which is centred in the hypothalamus organ in the brain, is the main mechanism that controls the timing of sleep, and is independent of the amount of preceding sleep or wakefulness. This internal clock is coordinated with the day-night / light-dark cycle over a 24-hour period, and regulates the body’s sleep patterns, feeding patterns, core body temperature, brain wave activity, cell regeneration, hormone production, and other biological activities. Even a long sleep might be ineffective (or at least inefficient) if it occurs at the “wrong” time of the circadian cycle. For more details see the following circadian rhythms – how sleep works.
But circadian rhythms alone are not sufficient to cause and regulate sleep. There is also an inbuilt propensity toward sleep-wake homeostasis, which is balanced against the circadian element. Sleep-wake homeostasis is an internal biochemical system that operates as a kind of timer or counter, generating a homeostatic sleep drive or pressure to sleep and regulating sleep intensity. It effectively reminds the body that it needs to sleep after a certain time, and it works quite intuitively: the longer we have been awake, the stronger the desire and need to sleep becomes, and the more the likelihood of falling asleep increases; the longer we have been asleep, the more the pressure to sleep dissipates, and the more the likelihood of awakening increases. For more details see the following sleep-wake homeostasis – how sleep works.
The interaction between the two processes can visualized graphically as follows:
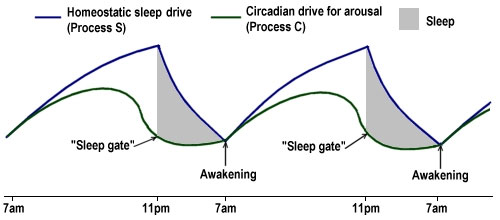
While homeostatic sleep drive typically increases throughout the day, effectively making a person more and more sleepy as the day goes on, it is countered and moderated by the circadian drive for arousal, at least until late evening, when the circadian clock slackens off its alerting system and begins sleep-inducing melatonin production instead. This opens the so-called “sleep gate” (marked by the point in the diagram above where the homeostatic sleep drive is at its greatest distance above the circadian drive for arousal). The exact way in which this occurs is still not fully understood, but the recent neuronal group theory of sleep theorizes that individual groups of neurons in the brain enter into a state of sleep after a certain threshold of activity has been reached, and that, once enough groups of neurons are in this sleep state, the whole organism falls asleep.
During the night, while sleep is actually being experienced, the homeostatic sleep drive rapidly dissipates, and circadian-regulated melatonin production continues. In the early morning, melatonin secretion stops and the circadian alerting system begins to increase its activity again. Eventually, the point is reached where the circadian drive for arousal begins to overcome the homeostatic sleep drive (marked by the point in the diagram above where the two curves meet), triggering awakening, and the process begins all over again.
While these are the two main processes involved, there are also other neurological mechanisms at work, including the suppression and re-activation of the everyday alertness signals of wakefulness, the switching between non-REM and REM sleep, the sleep inertia effect on waking, etc.
Other external factors, such as ambient temperature, meal times, naps, stress and exercise, also influence sleep times and durations, as of course can our own willed behaviour and daily schedules (e.g. work, school, alarm clocks). The practice of consciously following daily guidelines in order to regulate these external factors and ensure a more restful and effective sleep is known as "sleep hygiene", and can have at least a moderating or reinforcing effect on our underlying sleep patterns (see the separate section on Sleep Hygiene).
In addition, certain drugs (e.g. antihistamines, barbiturates, melatonin, certain psychoactive drugs, marijuana, alcohol, etc) act as sedatives, encouraging sleep, although care should be taken as some of these may also inhibit REM sleep and lead to disrupted sleep later, as a “rebound” effect occurs. For example, alcohol functions as a sedative initially and may help some people to get to sleep, but after a while it has a strong stimulant effect and actually disrupts sleep. On the other hand, stimulants (e.g. caffeine, amphetamines, ecstasy, cocaine, nicotine, etc) can be used to reduce or delay sleep, but they too have their own unfortunate side-effects and delayed rebound effects, and many are potentially addictive.